Pipeline
Pipeline by Disease
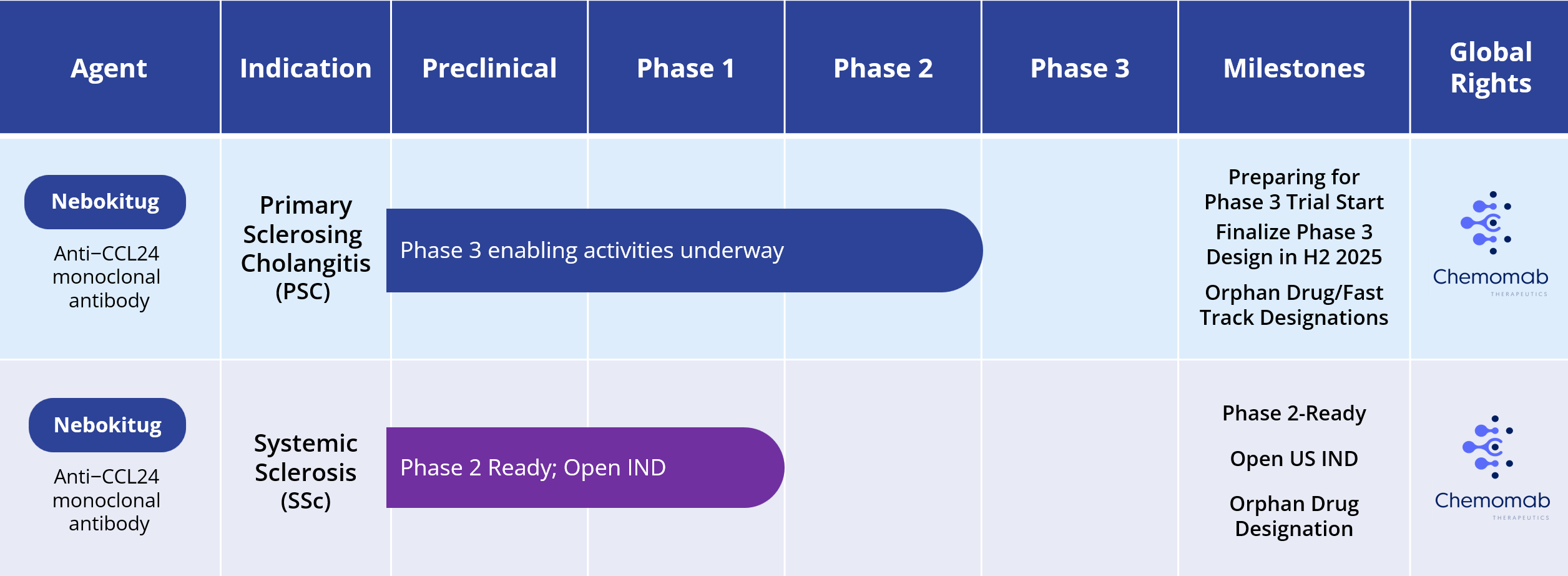
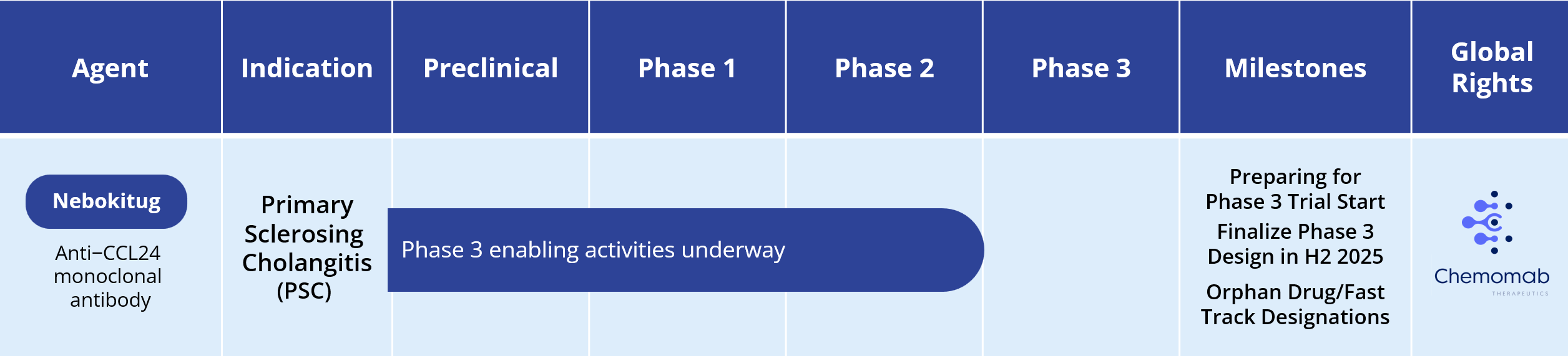
Primary Sclerosing Cholangitis (PSC)
Primary sclerosing cholangitis is a chronic progressive cholestatic disease of the liver characterized by inflammation and strictures of the biliary tree. The pathological hallmarks of PSC include injury to the integrity of the biliary ducts, retention of bile acids and intrahepatic inflammation and progressive liver fibrosis.
Disease Prevalence and Comorbidities
PSC is a rare disease, with a prevalence of 1 out of 10,000. It is more prevalent in men than women (2:1 ratio). There are an estimated 70,000 PSC patients in the major markets of the U.S., Europe and Japan. Peak age at onset of PSC is between 30 and 40 years. PSC patients frequently present with concomitant inflammatory bowel disease (up to 75%) and are at increased risk of developing hepatobiliary and colon cancers, which cause about half of all patient deaths. More than 50% of PSC patients need liver transplantation within 10–15 years of diagnosis, but in about 20% of these patients the disease reoccurs in the transplanted liver. PSC patients often have a poor quality of life, with fatigue, severe itch, jaundice, abdominal pain and hospitalizations making normal life difficult. Median transplant-free survival is 10-20 years.
Current Treatment
There is no approved drug for treating PSC. Current treatment focuses on reductions of cholestasis and management of disease manifestations and complications (e.g., fatigue, pruritis).
To learn more about how CM-101 may interrupt the fibro-inflammatory vicious cycle believed to drive disease progression in PSC, see our Mechanism of Action (MOA) video at: MOA video.
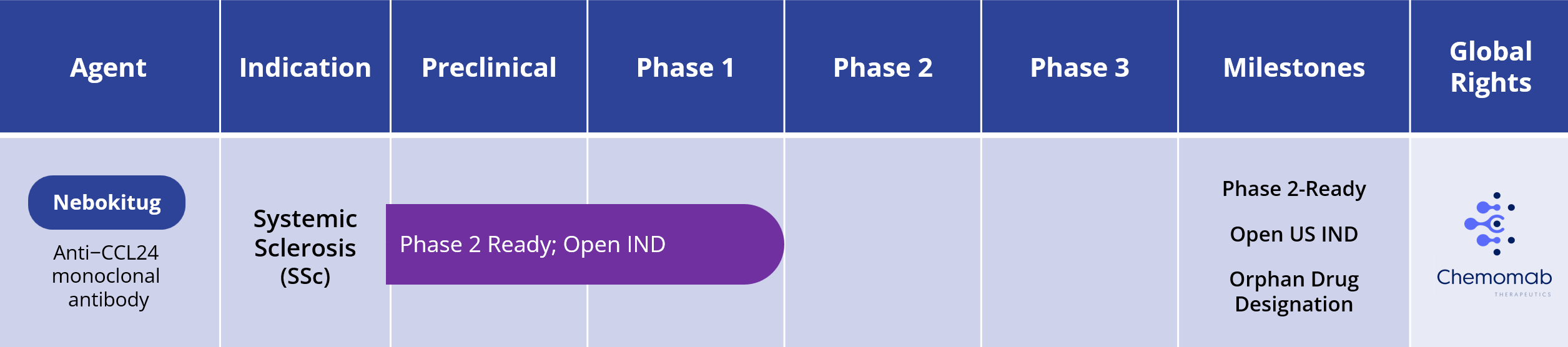
Systemic Sclerosis (SSc)
Systemic sclerosis, also called scleroderma, is a rare, chronic progressive autoimmune disease that is characterized by chronic inflammation, fibrosis of the skin and internal organs and vascular damage. SSc can involve multiple internal body organs and the symptoms of the disease vary depending on the affected organs and extent of damage.
Disease Prevalence and Comorbidities
Peak age at onset of SSc is between 20 and 50 years, and it is more prevalent in women compared to men, at a 3:1 ratio. There are two forms of SSc:
- Limited cutaneous SSc, which is a slowly progressive disease with skin involvement restricted to the limbs and characterized by delayed-onset organ damage
- Diffuse cutaneous SSc, which is a more aggressive form of the disease that presents a widespread skin damage and risks rapid-onset organ damage, including effects on the kidneys, heart, lungs and gastrointestinal tract.
SSc is considered rare, with an estimated prevalence of approximately 1 in 10,000 and an estimated 170,000 patients in the seven major markets of the U.S., Europe and Japan. The prognosis varies significantly among patients and is correlated to the extent of organ damage. SSc has high morbidity and the highest mortality rate among the systemic rheumatic diseases, with a median survival of about 10 years. Pulmonary damage is the main cause of death but patients face many painful and debilitating symptoms and disabilities from the varied manifestations of SSc.
Current Treatment Options
One therapy is approved for slowing the rate of decline in pulmonary functions in patients with SSc associated with interstitial lung disease (SSc-ILD). Currently, there is no therapy approved for SSc beyond this manifestation. Current treatment is therefore directed at suppression of the immune system or management of specific disease manifestations (e.g., digital ulcers, pulmonary hypertension).
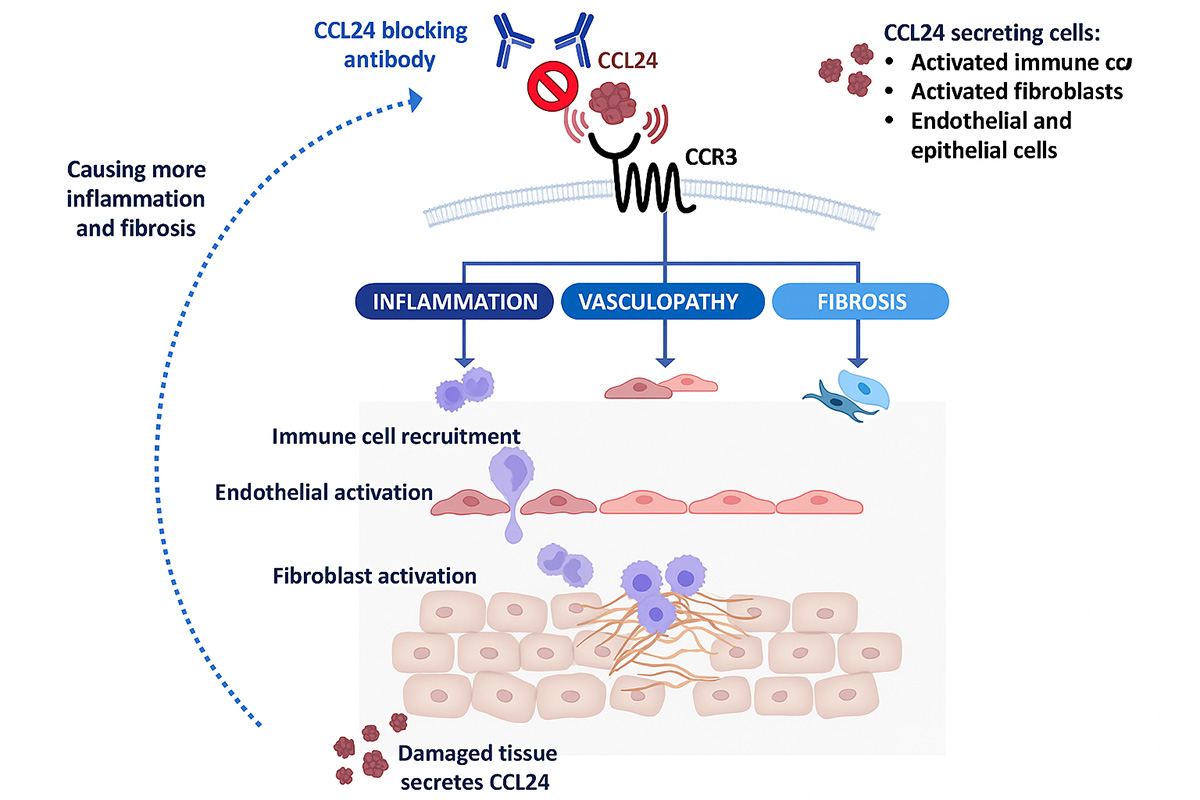
CCL24 Involvement in SSc Pathology
SSc is considered rare, with an estimated prevalence of approximately 1 in 10,000 and an estimated 170,000 patients in the seven major markets of the U.S., Europe and Japan. The prognosis varies significantly among patients and is correlated to the extent of organ damage. SSc has high morbidity and the highest mortality rate among the systemic rheumatic diseases, with a median survival of about 10 years. Pulmonary damage is the main cause of death but patients face many painful and debilitating symptoms and disabilities from the varied manifestations of SSc.
Levy et al Rambam Maimonides Medical Journal 2023
The Phase 2 SPRING trial proof-of-concept data expanded the rationale for advancing nebokitug in systemic sclerosis. It is the first clinical validation of nebokitug’s mechanism of action and its anti-inflammatory and anti-fibrotic effects, including beneficial changes in PRO-C3, ELF and inflammatory cytokines, which are well-established indicators of SSc disease activity. The SPRING trial also reinforced the extensive preclinical evidence of nebokitug’s therapeutic potential in SSc, which includes animal studies, in-vitro and in-vivo experiments, and patient sample and clinical studies that support CCL24 blockade as a therapeutic strategy. For example, CCL24 blockade inhibits collagen deposition, reduces dermal thickness and decreases immune cell infiltration into the lung. Importantly, a number of studies have shown that high CCL24 levels are linked to increased fibrosis biomarkers and decreased lung function, and they can actually predict mortality in SSc patients. Additionally, treatment with nebokitug demonstrated strong and rapid reductions of inflammatory biomarkers in hospitalized patients with acute lung Injury.
Nebokitug has received Orphan Drug designation for systemic sclerosis is the U.S. and European Union. Chemomab has an open U.S. IND to conduct a Phase 2 clinical trial of nebokitug in patients with systemic sclerosis.